This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison.
What are Protein domains?
Distinct functional units in a protein are known as domains. The domains of a protein are each responsible for a specific function that work together to contribute to the overall role of the protein as a whole. Protein domains exist in a multitude of biological contexts, as well as similar domains can serve different functions in other proteins (1). Domains of the FLNB protein and their function can be seen below.
What domains make up the FLNB protein?
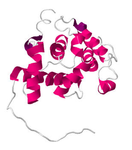
The FLNB protein is fairly long, 2633 amino acids in length. FLNB consists of 2 CH (calponin homology) domains and 23-24 Filamin repeat domains (Figure 1-3). The domain structure of FLNB across different databases is consistent, with some variation in the number of Filamin repeats seen. How these 2 types of domains contribute to the overall role of the protein is explained below. Figure 1 was generated from SMART by imputing FLNB un-annotated protein sequence (file below) and having "genomic" smart mode selected. Figure 2 was generated from Pfam by clicking on "view a sequence" and entering in the search box "FLNB_HUMAN". Figure 3 was generated from PROSITE by imputing FLNB un- annotated protein sequence (file below).
How well conserved is FLNB across species?
The FLNB protein is fairly conserved across homologous species as shown in Figure 4. The protein structure is very conserved among mammalian species (human, chimp, mouse) which have very close amino acid number and the same number of CH and filamin domains. Across all species the CH domain is very conserved where all species represented having 2 CH domains, except the plant Arabidopsis which has 3 CH domains, which make up what's called the Actin Binding Domain of the FLNB protein. There chimp and mouse both have 24 filamin domains like humans The zebra fish have 16, Drosophila have 19, nematode have 15 , and plants have 0 filamin domain repeats. Note the filamin-like domain seen in zebra fish is an immunoglobulin like domain structure which hasn't been classified (2).
To determine the domain structure of each homologous species shown in Figure 4, each species un-annotated filamin B protein amino acid sequence was seperatley copied into SMART. Make sure that the "genomic" smart mode is selected, and the protein domains for that species will appear. I reconstructed these results into one image for easy comparison. All the species protein amino acid sequences can be found at the bottom of the FLNB Homology page by clicking on their specific FASTA link.
To determine the domain structure of each homologous species shown in Figure 4, each species un-annotated filamin B protein amino acid sequence was seperatley copied into SMART. Make sure that the "genomic" smart mode is selected, and the protein domains for that species will appear. I reconstructed these results into one image for easy comparison. All the species protein amino acid sequences can be found at the bottom of the FLNB Homology page by clicking on their specific FASTA link.
What is the function of CH domain?
|
What is the function of the Filamin domain?
|
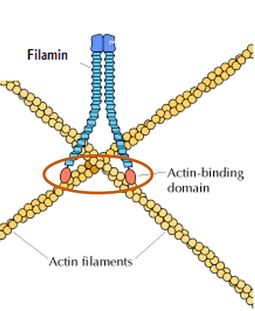
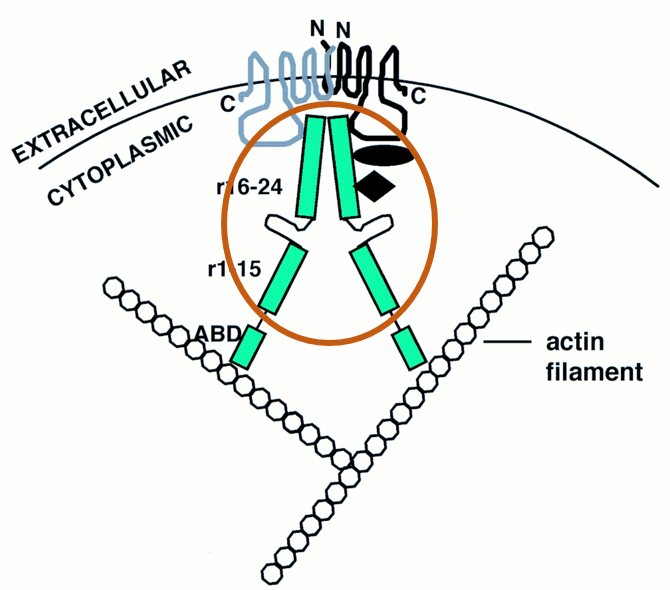
The rod-shaped filamin domain (Figure 8), located at the C-terminal, of the human FLNB protein is repeated 23-24 times. Repeats of the filamin domain are common and are constructed of a 100-residue motif that is glycine and proline rich (5). Filamins function is also involved in actin binding cytoskeleton formation (Figure 7) as well (5). Filamin interacts with plasma membrane proteins bringing the actin filaments attached by the ABD close to the cytoplasmic edge of the plasma membrane(Figure 9). This allows the actin filaments to surround the outer edges of the whole cell. |
Analysis:
Now that we understand the components that make up the FLNB protein and their roles separately and as a whole, we can then start to figure out what might be the reason for the differences in protein structure in non-mammalian species. As observed in Figure 4 mammalian species, such as the chimp, mouse and human, FLNB protein structure is very conserved. This makes sense because we know that FLNB protein is involved in cartilage and bone development and mammals have bones. But, there is some variation in the Filamin B protein structure in zebra fish, which we know is a boney fish. There is also much variation in the other non-mammalian species shown in Figure 4, especially in the plant homolog of filamin B, fimbrin-like protein. As you can see the plant protein has 3 CH domains and zero filamin domains. This is because plant cells contain actin, which explains why the CH domains are seen in plants, but the actin connects to their membranes in another way, which is why filamin is not seen in its protein structure (6). Plants contain a unique domain, NET1A , specific to plants that is involved in actin-binding and connects actin-filaments not only to the plasma membrane but through their cell walls (6). In nematodes the FLN-1 protein is involved in brood size (7). But, when is comes to trying to determine the function of filamin-B in zebra fish and filamin-1 in Drosophila nothing is known. It would be interesting to further explore the role FLNB protein homologs in non-mammalian species, specifically in the fruit fly since it doesn't have bones, as well as in the zebra fish for there is variation in the protein even though they have bones and no research has been done to determine why.
| SMART and PROSITE: FLNB protein input sequence.docx | |
| File Size: | 14 kb |
| File Type: | docx |
References:
1.) "Introduction to Protein Classification at the EBI: What Are Protein Domains?" European Bioinformatics Institute, 2014. Web. May 10, 2014. http://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
2.) "SMART: IG-like". Web. April 12, 2014. http://smart.embl-heidelberg.de/smart/do_annotation.pl?DOMAIN=IG_like&START=1193&END=1246&E_VALUE=11.7235843947643&TYPE=SMART&BLAST=PGRIAVKLTNSEGIPVDNLRVEDKGNCIYAVHYVPPKAGSVLTCQVKFSEVEVP
3.) "SMART: CH Domain Annotation." Web. February 25, 2014. http://smart.embl-heidelberg.de/smart/do_annotation.pl?DOMAIN=CH&START=141&END=237&E_VALUE=1.31513952069162e-19&TYPE=SMART&BLAST=PKQRLLGWIQNKIPYLPITNFNQNWQDGKALGALVDSCAPGLCPDWESWDPQKPVDNAREAMQQADDWLGVPQVITPEEIIHPDVDEHSVMTYLSQF
4.) Sawyer, Gregory, M., et. al., (2009). "Disease-associated Substitutions in the Filamin B Actin Binding Domain Confer Enhanced Actin Binding Affinity in the Absence of Major Structural Disturbance: Insights from the Crystal Structures of Filamin B Actin Binding Domains". Journal of Molecular Biology, 390(5), doi: 10.1016/j.jmb.2009.06.009
5.) "SMART: IG_FLMN Domain Annotation." Web. February 25, 2014. http://smart.embl-heidelberg.de/smart/do_annotation.pl?DOMAIN=IG_FLMN&START=253&END=350&E_VALUE=5.11222530099724e-34&TYPE=SMART&BLAST=NPKKARAYGRGIEPTGNMVKQPAKFTVDTISAGQGDVMVFVEDPEGNKEEAQVTPDSDKNKTYSVEYLPKVTGLHKVTVLFAGQHISKSPFEVSVDKA
6.) Clive Lloyd. (2012). "Plant Actin: Trying to Connect You". Current Biology, 22(17), doi: http://dx.doi.org/10.1016/j.cub.2012.06.071
7.) Kovacevic, I., Cram, EJ, (2010). "FLN-1/filamin is required for maintenance of actin and exit of fertilized oocytes from the spermatheca in C. elegans." Developmental Biology, 347(2), doi: 10.1016/j.ydbio.2010.08.005
2.) "SMART: IG-like". Web. April 12, 2014. http://smart.embl-heidelberg.de/smart/do_annotation.pl?DOMAIN=IG_like&START=1193&END=1246&E_VALUE=11.7235843947643&TYPE=SMART&BLAST=PGRIAVKLTNSEGIPVDNLRVEDKGNCIYAVHYVPPKAGSVLTCQVKFSEVEVP
3.) "SMART: CH Domain Annotation." Web. February 25, 2014. http://smart.embl-heidelberg.de/smart/do_annotation.pl?DOMAIN=CH&START=141&END=237&E_VALUE=1.31513952069162e-19&TYPE=SMART&BLAST=PKQRLLGWIQNKIPYLPITNFNQNWQDGKALGALVDSCAPGLCPDWESWDPQKPVDNAREAMQQADDWLGVPQVITPEEIIHPDVDEHSVMTYLSQF
4.) Sawyer, Gregory, M., et. al., (2009). "Disease-associated Substitutions in the Filamin B Actin Binding Domain Confer Enhanced Actin Binding Affinity in the Absence of Major Structural Disturbance: Insights from the Crystal Structures of Filamin B Actin Binding Domains". Journal of Molecular Biology, 390(5), doi: 10.1016/j.jmb.2009.06.009
5.) "SMART: IG_FLMN Domain Annotation." Web. February 25, 2014. http://smart.embl-heidelberg.de/smart/do_annotation.pl?DOMAIN=IG_FLMN&START=253&END=350&E_VALUE=5.11222530099724e-34&TYPE=SMART&BLAST=NPKKARAYGRGIEPTGNMVKQPAKFTVDTISAGQGDVMVFVEDPEGNKEEAQVTPDSDKNKTYSVEYLPKVTGLHKVTVLFAGQHISKSPFEVSVDKA
6.) Clive Lloyd. (2012). "Plant Actin: Trying to Connect You". Current Biology, 22(17), doi: http://dx.doi.org/10.1016/j.cub.2012.06.071
7.) Kovacevic, I., Cram, EJ, (2010). "FLN-1/filamin is required for maintenance of actin and exit of fertilized oocytes from the spermatheca in C. elegans." Developmental Biology, 347(2), doi: 10.1016/j.ydbio.2010.08.005